Is Vaccinating Kids 6 Months Up To 5 Years Against COVID-19 Safe?
Pfizer seeks EUA For Young Kids
Pfizer could be submitting a request to the FDA for approval of their 2 dose COVID-19 mRNA vaccine for those 6 months up to 5 years old as early as today. Currently it is approved for EUA (emergency use authorization) for those 5 years old and up. This publication will shed light on 2 areas.
The available data from Pfizer’s phase 1/2/3 vaccine clinical trial for kids 6 months to 5 years old
And If vaccinating kids 6 months old up to 5 years is evidence based
The data
Before Pfizer can apply for EUA through the FDA, appropriate clinical trials have to be completed that show efficacy and safety for the cohort which will receive the intervention. Generally speaking, that means the vaccine is given to one group, while another group is a control receiving only a placebo. All that is applied in a blinded context where individuals are prevented from knowing certain information which decreases bias in the trial. Now that the table has been set, salient data points summarizing Pfizer’s phase 1/2/3 clinical trial for kids 6 months up to 12 years will be listed as bullet points below.
Originally clinical trial included 4,500 total children, now 8,300 aged 6 months to under 12 years
Participants from USA, Finland, Poland & Spain
Total of (n=4,500) children derived from 90 clinical sites
3 age groups were given the treatment: 6 months- 2 years, 2 years - under 5 years, and 5 years- 12 years
Ages 6 months - 5 years received 2 smaller 3 mcg doses 21 days apart
Ages 5-12 received 2 larger 10 mcg doses 21 days apart
Results so far: Vaccine didn’t meet the low bar of non inferiority on antibody titers for those aged 2-5 (in other words, antibody levels conferred from vaccination were arguably too low to generate protection)
To gain approval of dose 1 & 2, a 3rd dose will also need to prove efficacious in this study because the antibody response from dose 1 & 2 was insufficient
Is vaccinating children 6 months up to 5 years old evidence based?
If vaccinating children from 6 months up to 5 years was evidenced based, 2 major factors would need to need to be true. First, the risk of severe disease, or death for said cohort would need to be high, especially from Omicron compared to Delta. Second, the benefits would need to outweigh the harms.
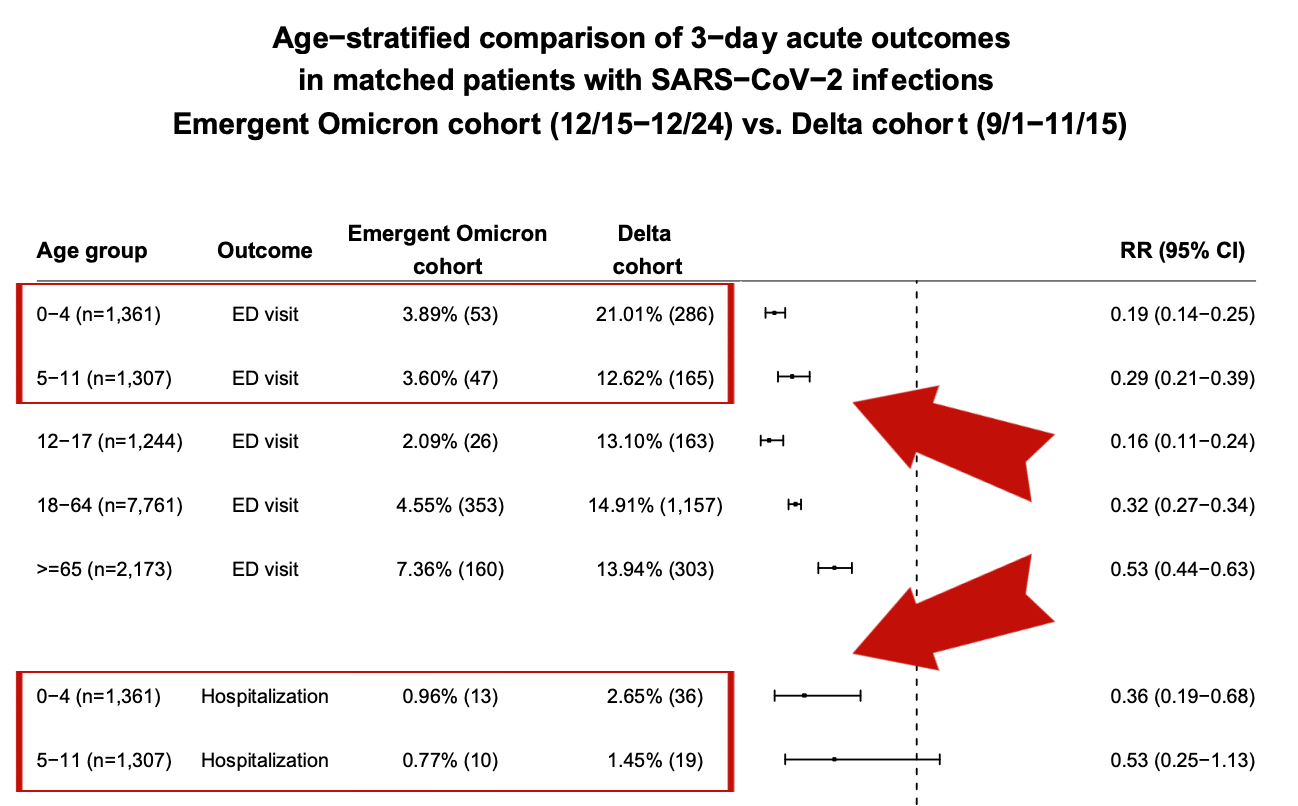
Regarding the first factor, the data (here, here, and here) clearly suggest that although a sizable amount of children 4 and under presented to hospitals with Omicron, they experienced less severe disease than the same age group with Delta overall (see FIG. 1 📸). As you can see from the first red rectangle below, data from this study shows that compared to Delta, children between the ages of 0-4 have a nearly +/- 80% reduced risk of being admitted to the ED from Omicron compared to Delta. Also, the same age bracket has a +/- 75% reduced risk of hospitalization from Omicron compared to Delta. Moving down to the second red rectangle, risk of hospitalization is nearly halved. The point is, severity of disease is less with Omicron compared to Delta for kids. Keep scrolling to read more 👇.
FIG. 1 📸
From the same study referenced in the picture above, data in the image below (FIG. 2 📸) is stratified to show mechanical ventilation from Omicron in sufferers 5 and under compared to those with Delta (you can this study here). If you allow your eyes to find the next red rectangle below, you will see that mechanical ventilation was reduced overall by nearly +/- 80%. In other words, children do very well with the new variant and hospitalizations were likely a result of anxious parents, and respiratory/other symptoms that resolved within 3 days or so. Sadly, it is true some required ventilation, but fortunately there were no deaths recorded. Keep scrolling 👇.
FIG. 2 📸
Interestingly, another study showed that infections/cases were higher in kids 1-9 but the good news is, the previously explained data showed that infections ended up being mild and short lived. See FIG. 3 📸 below (SGTF in the chart below represent those with Omicron S- gene target failure- Conversely, NO SGTF represents Delta). That said, it is true many people are fixated on increased case numbers. However, those Omicron cases are yielding less severe disease than Delta and are therefore a poor metric to validate vaccination. All and all, from the most current data, children don’t seem to suffer severe disease from Omicron very frequently so for this reason alone vaccination can’t be justified.
You can’t make a child healthier, but you can certainly give them side effects from vaccinating. Keep scrolling 👇.
FIG. 3 📸
Regarding the second factor. The benefits of vaccination need to outweigh the harms. Regrettably, that is unlikely to happen for a few reasons.
You see, this cohort does very well with the virus, and symptoms from Omicron are mild in nature, so any adverse event experienced from vaccination will likely make it more harmful than efficacious. To be specific, even a singular unit of harm per 100,000 would surely reveal the intervention to be more detrimental than simply facing the virus. Unfortunately, the data will reveal at least that and more. But there’s another problem.
The Pfizer clinical trial of only 4,500 children is underpowered and will not accurately reveal the true extent of severe adverse events. You can not appropriately extrapolate vaccine side effects from such a small number.
Also, let’s not forget the 3 mcg dose of this vaccine didn’t even meet the low bar of non inferiority on antibody titers for those aged 2-5. That’s why this trial was extended by adding a booster dose. Essentially, the trial failed to meet it’s primary endpoint- adequate antibody titers.
Considering the facts, vaccinating children 6 months up to 5 years will surely do more harm than good. That being the case, Omicron is less severe in nature so COVID-19 may need to be approached as a childhood disease that younger kids have a brush with instead of vaccinating against.
The takeaway
When Pfizer releases further details from their phase 1/2/3 clinical trial regarding vaccinating the age groups listed above, the results based on neutralizing antibody titers may somewhat justify the intervention. However, again, antibodies conferred by vaccination can not improve upon the nearly perfect immune systems of children aged 6 months up to 5 years by any order of magnitude which makes this intervention unavailing. Also, the harms suffered from adverse events will likely outweigh the good making this vaccine non-efficacious overall for this particular cohort.
LETS CONNECT:






The long term effect of the vaccines will probably be, amongst others, an altered, weakened immune system. Kids will become chronic big pharma clients.