New "NeoCoV" Virus Is Not More Lethal Or Highly Transmissible In Humans
The media is focusing on a “new” category of coronavirus linked to MERS (Middle East respiratory syndrome) which could potentially cause major harm throughout the global population. Essentially, their opinion is drawn from a new study out of Wuhan China inferring that a bat virus named “NeoCov" is only one mutation away from being able to infect humans and cause severe disease. That evidenced by in-vitro lab results. However, zoonotic transmission to humans is a farce. So in this publication 3 areas will be examined:
The history, and pathology of COVID-19
The history and pathology of MERS-CoV, and how the data from this new “NeoCov” study doesn’t mean the virus will hop to humans
History and pathology of COVID-19
SARS-CoV-2 is a virus that causes the disease COVID-19. It first appeared in late 2019 in Wuhan China. Soon after in January 2020, the WHO declared the virus a public health emergency of international concern. Following that, in March of 2020 SARS-CoV-2 was deemed a global pandemic.
Next order of business. The novel SARS-CoV-2 virus infects host cells by a specific mechanism. It is important to note that this method has been used by the virus since the original variant up until Delta. However, Omicron infects cells differently and is not applicable to this discussion. To learn more about Omicron, tap here.
As a prelude to the next paragraph, please keep in mind ACE2 receptors play a major role in modulating blood sugar, blood vessel integrity, inflammation, and lung damage. So it could be said, in the absence of appropriate ACE2 functioning, cells are at high risk for damage.
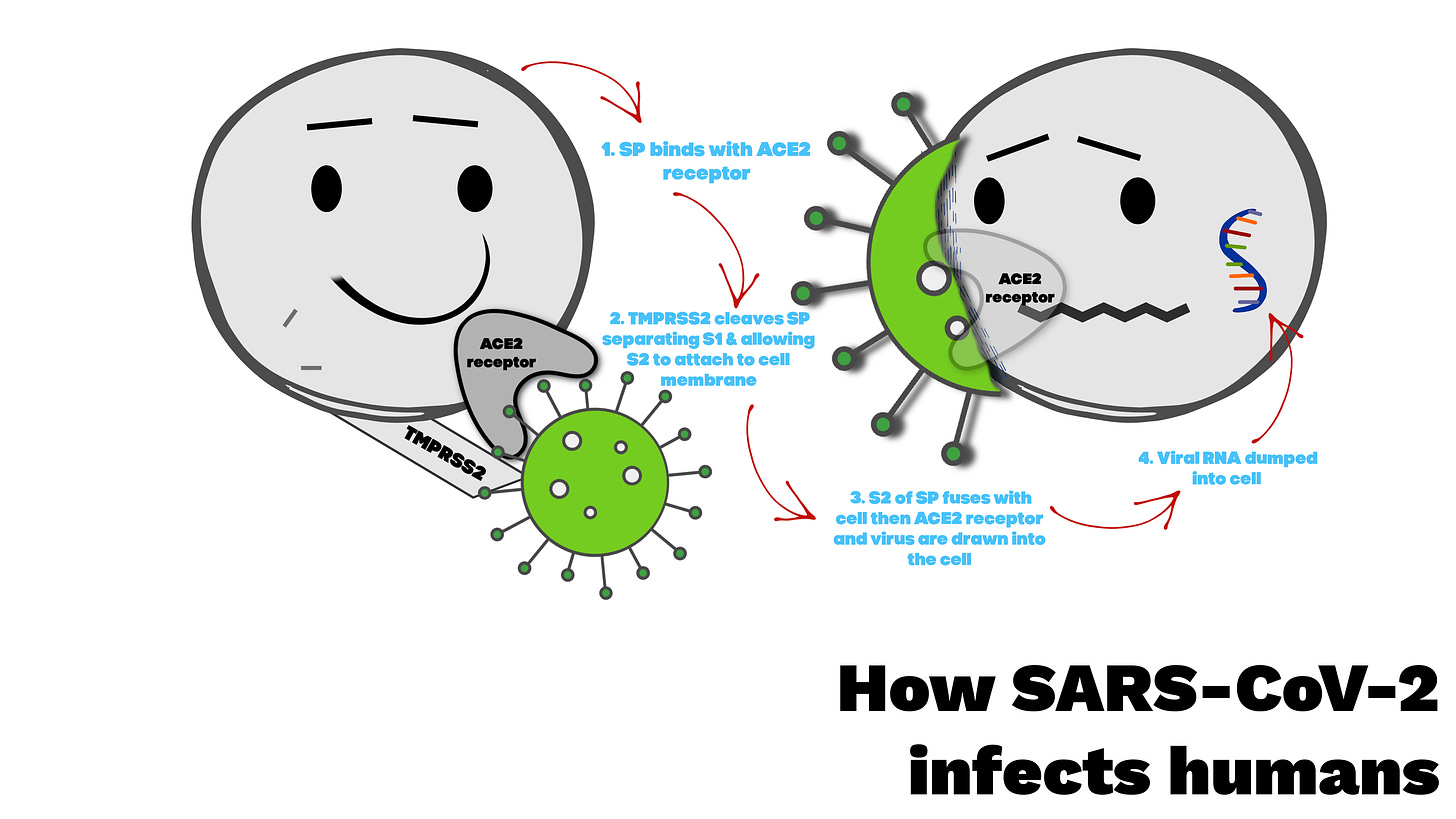
The problem. Spike protein from SARS-CoV-2 connects to the ACE2 receptor instead of the ACE enzyme, and that begins a cascade of inflammatory events in places like the lungs, heart, kidneys, testes, and oral mucosa (FIG. 1 📸). The process goes like this. Starting with “#1” from the picture below, the virus finds its way to an ACE2 receptor on a human cell. Next, a human membrane enzyme called TMPRSS2 cleaves, or separates the spike protein from the virus into 2 parts, S1 & S2. The remaining truncated S2 segment fuses with the host membrane allowing the virus to dump its RNA into the cell and down regulate the ACE2 receptor which hastens lung, cardiac, kidney, gastro damage. After that, the infected cell churns out its progeny which then go onto infect other cells.
FIG. 1 📸
The resulting pathology in lung, cardiac, kidney, and sexual tissue is due to an imbalance of homeostasis within the cells caused by down regulation of ACE2 receptors that then dysregulate RAS (renin angiotensin system). RAS if unchecked, inadvertently produces serious metabolic, cardiac, and pulmonary side effects. Particularly, SARS-COV-2 has a proclivity to convert ANGI enzyme to ANGII. In doing that, expressed ANG receptors from ANGII enzymes can increase thrombotic activity (blood clotting) via endothelial damage, disrupt internal structures of blood vessels and capillaries in the lungs and heart which alter oxygen exchange, and of course hasten metabolic problems like high blood sugar.
History and pathology of MERS-CoV
To start, please understand that MERS-CoV is the virus that causes the disease MERS (Middle Eastern respiratory syndrome). It can produce serious symptoms and spread at an alarming rate. Luckily, this is no longer experienced in the USA.
In June of 2012 the first cases of MERS-CoV were reported in Saudi Arabia. Soon after, the virus spread to places like America, Europe, and Asia. Unfortunately, the hardest hit were Middle Eastern countries, hence the name Middle Eastern respiratory syndrome. Shortly after in 2015 there was another outbreak in South Korea. Fortunately, it is now under control and endemic to the Middle East. As for the United States, there were only 2 reported cases in 2014.
Next, if one were to face the virus, symptoms would include severe respiratory distress, nausea, fever, and diarrhea that could lead to death. For some who experienced the blunt end of the disease, symptoms carried on for years after the acute phase resolved- that strikingly similar to long COVID. Sadly, the mortality rate for MERS was very high around 30%.
Now, you should know MERS-CoV coronavirus affects bats, camels, and humans. It invades by first attaching to specific receptors like S1-CTD, ACE2, DPP4, and APN (FIG. 2 📸). Also, infection occurs in a somewhat similar fashion to what was previously explained in FIG. 1 📸. Notably, MERS-CoV mainly uses DPP4 receptors in animals. However, NeoCoV, a subgroup of MERS-COV found in Africa uses ACE2 receptors to gain entry and infect their host.
FIG. 2 📸
Evaluation of the “NeoCoV” study
The authors of the above mentioned study state that historically “NeoCoV” and PDF-2180-CoV (a similar virus), have an affinity for the same bat ACE2 receptors and therefore, NeoCoV may infect humans by using their ACE2 receptors. Regrettably, the causal link doesn’t hold water. Here’s why.
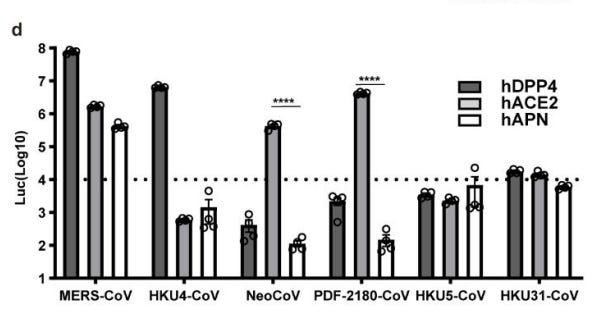
To start, scientists in this study engineered human 293T cells that expressed ACE2 receptors, while measuring the entry of merbecoviruses with a pseudovirus entry assay to estimate the ability of NeoCoV and PDF-2180-CoV viruses to infect the engineered human 293T cells with attached ACE2 receptors. Said differently, scientists took lab engineered human cells, then attached ACE2 receptors, and tried to infect them with both NeoCoV, and PDF-2180-CoV viruses. Following that, the results were confirmed with a pseudovirus entry assay- or test. To the scientists surprise, both viruses infected the lab engineered human 293T cells. To give you a visual, check out FIG. 3 📸 below. As you can see, NeoCoV had a very high cell entry efficiency rate, check out the hACE2 (human ACE2) bar. All and all, from that data, authors suggested the possibility of NeoCoV infecting human cells. However, in the same breath they implied it was unlikely to happen anytime soon.
Interestingly, they are assuming a causal relationship between a virus infecting an animal, and a virus infecting a human solely based on lab data. More specifically, entry of a virus into a bat, and entry of the same virus into a lab engineered human cell in-vitro. Sadly, that doesn’t translate to the virus infecting a human. To explain, the human body is much more complex than an isolated lab cell where all variables are accounted for. In humans, you can’t account for a perfect cellular environment like in a lab, there are often imperfect conditions unbalancing the body like disease, and aging. That said, the data is again interesting, but nothing more than that.
It should also be stated that experiments like this are not often replicated well. For instance, the same authors could go through the exact process again and end with completely different outcomes. So you understand, this is why we don’t use in-vitro data to justify anything in clinical practice. Conversely, randomized controlled trails are the gold standard.
So is NeoCoV something to be worried about? No. However, it seems the media is attempting to stir up panic by distorting the data. Nonetheless, now that you’ve read this you understand why NeoCoV is a no-go.
FIG. 3 📸
LETS CONNECT: