Omicron Will Likely End The Pandemic
At this point, Omicron makes up over 95% of cases in the United States. That means, Delta is no longer dominant. Moreover, for Delta to no longer be as influential, Omicron had to have an edge over it competitor. Fortunately for all, that edge made the variant less virulent, allowing it to survive longer in its host, and produce less severe symptoms.
You see, a virus wants to survive, and a smart one mutates into something less harmful to the environment it inhabits. After all, a virus can’t proliferate and pass on its progeny if it severely disables, or kills its host. That said, the nearly 50 mutations in Omicron made it more transmissible, but less deadly.
As evidence of the previously stated, a new study out of the UK shows Omicrons new mechanism which allows it to infect cells easier, leading to rapid spread throughout the population but with less severity of symptoms and death overall. As a side note, check out my previous article showing how Omicron confers a 53% reduced risk of any subsequent hospitalization, 53% reduced risk of symptomatic hospitalization, 74% reduced risk of ICU admission, and 91% reduced risk of death compared to Delta.
Now with all of that out of the way, understand that for a virus to become endemic 3 important things need to happen. One, there needs to be multiple animal reservoirs. Two, the virus must become less virulent. Three, the virus needs to be constantly present in the population. We are now seeing all three.
Considering the evidence, Omicron will likely turn the pandemic, endemic. Anyways, the data will now be explored relating to how Omicron is more transmissible yet less deadly.
How Delta infects cells (📸 FIG. 1)
First you need a little history on how Delta infects cells before we dive into Omicron. Let it be known, the gray circles below are cells, and the green spheres are SARS-CoV-2, the novel virus that causes the disease called COVID-19. Just so you know, all numbered points below will correspond with numbers within the associated images/slides in blue text.
First, spike protein (SP) from the virus attaches with an ACE2 (angiotensin converting enzyme) receptor on a human cell
*keep in mind SP is made up of 2 segments, S1 and S2. S1 is the top circular tip seen on the virus, and S2 is what connects that circular tip to the body. You could also call S2 the trunk
Then, a protease from our cell called transmembrane protease serine 2 (TMPR22s) cleaves, or cuts, the viral SP which separates the S1 and S2 segments. What’s left is a truncated S2 segment (green) that can now attach to the cell membrane (in gray)
The remaining S2 segment of SP fuses with the cell. After, both ACE2 receptor and virus are drawn into the cell. Keep in mind, at this time the ACE2 receptor being drawn into the cell is down-regulated which begins a cascade of inflammatory events. In other words, this is when the symptoms can begin to manifest
At the same time, the virus dumps it's RNA, or instructions into to the host. Those instructions will program the host to produce more virus. Essentially, once infected, it then becomes a virus producing machine yielding new progeny that will go on to infect even more health cells in the body. So on and so forth. As that happens, viral load increases. It is at this time, an individual could begin testing positive.
Syncytia formation (📸 FIG. 2)
This is a hallmark of Delta, it’s called syncytia formation. Basically this is when the virus forces little fibers from lung cells called cilia to tangle together which then causes fusion of pseudostratified columnar ciliated epithelium lung tissue. In other words, the viruses spike protein gets into deep lung tissue, causes tangles, then fuses lung cells making them unable to oxygenate the body. Importantly, this mechanism is what causes severe deep lung disease in patients with Delta. You’ve heard of symptoms like shortness of breath, and respiratory failure. All that a result of the aforementioned process. Now, two important points:
Omicron doesn’t cause as much syncytia formation (damage) as Delta ✅
That’s because TMPRSS2 isn’t priming SP in Omicron (as previously explained for Delta). Omicron goes through a different process to enter/infect cells ✅
Delta in deep lung tissue (📸 FIG. 3)
To continue what was explained in the previous slide, we will now look in detail at the mechanism Delta uses which causes severe lung disease. Of course, we’ll get to Omicron soon.
To start, Delta has an affinity for lung tissue, so it finds its way to the lungs and attaches with little hairs on top of cells called cilia. Unfortunately, that process ends up fusing cilia, and neighboring membranes together. Moreover, it causes cytokines to be released by the affected cells. To be specific, cytokines are little chemical proteins that alert the immune system there’s a problem.
As a result, big cells like macrophages and dendritic cells sense those cytokines (chemical messages) and make their way over to the affected area and help break down the affected cells which initially sent the alert.
Regrettably, help from macrophages and dendritic cells cause a substantial amount of damage that the body can’t repair fast enough. So, as you could understand when the lungs are in good measure breaking down faster than they can be rebuilt, disease happens. Of course, the disease is named COVID-19.
As a side note, the reason this process happens frequently in deep lung tissue is because SARS-CoV-2 LOVES ACE2 receptors + ciliated cells, and there are an abundance of them within the lung.
It is important to note, Omicron predominantly affects the upper airway, not deep lung tissue which is why disease is less severe*
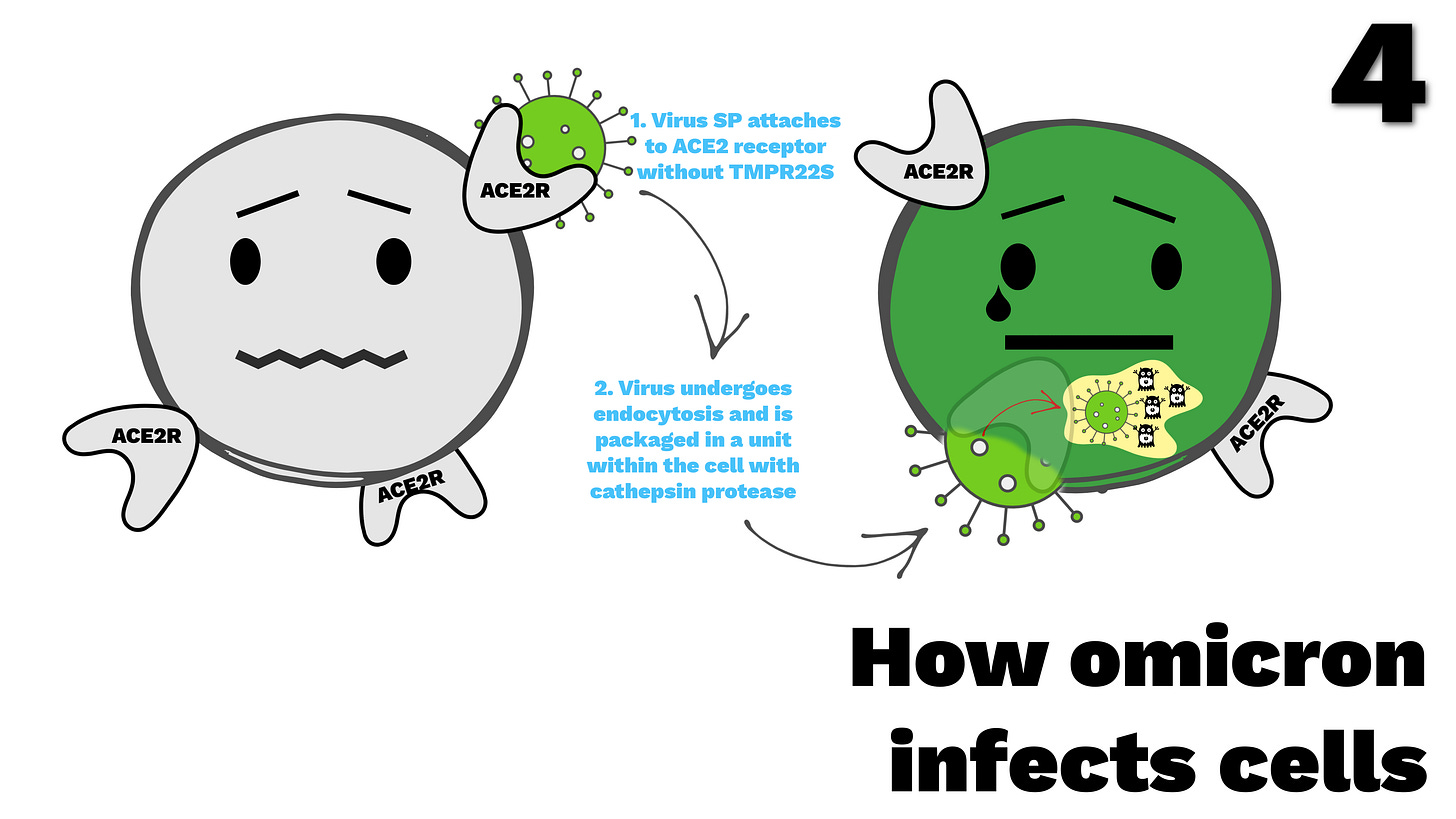
How Omicron infects cells (📸 FIG. 4)
As a prelude to the slide below, please understand we will now begin examining the newest data on Omicron’s pathogenicity illuminating how the variant causes less severe symptoms (references here, here, and here).
Omicron finds its way to the same ACE2 receptor that Delta uses. The difference, after connecting with the ACE2 receptor, Omicron’s SP doesn’t need to be cleaved, or cut, by the human membrane protease TMPRSS2 like Delta.
Instead, to infect, Omicron enters the human cell cytoplasm by endocytosis. Basically, that means the virus in its near full and unadulterated form, ends up being surrounded by a protective membrane by the same cell it ends up entering. As that happens, many human cathepsin proteases get trapped inside the protective membrane with it (see yellow endocytotic vessel with virus and cathepsin together below on the right).
Cathepsin liberating Omicron (📸 FIG. 5)
This is a very important part because once Omicron is freed from its vessel, it begins causing problems. As previously explained, cathepsin is a protease that will in this case, help liberate Omicron from that yellow vessel below. After liberated, the virus can rapidly produce progeny, which will in turn increase viral load, which then brings on more symptoms. Here is the sequence of events.
A lysosome senses the newly entered endocyte and sees it as a threat. It then begins breaking down the threat. That happens by the lysosome spilling enzymes on the yellow viral package holding both the virus and cathepsin. That process digests the wall protecting them which enables cathepsin and the virus to become liberated from the vessel. This process lowers the the PH (which means increases the acidity) inside the cell.
Sadly, the same increase in acidity that usual annihilates invaders, helps Omicron in this case. More specifically, it activates cathepsins.
Finally, after cathepsin is activated it benefits the virus by priming it (preparing it) within the cell, instead of outside (as previously explained). Such a process makes it even easier for the virus to infect subsequent cells because the virus skips priming from TMPRSS2. Essentially, the virus leaves the cell pre-primed, immediately ready to fuse and infect the next cell.
Spike proteins (📸 FIG. 6)
The images below denote how researchers in this study proved Omicron was entering the cell differently compared to Delta hence making it more infectious, but less virulent. To explain, they took spike protein from 1.) the original strain, 2.) Alpha variant, 3.) Delta variant, 4.) Omicron, and 4.) pangolins (ant eaters). After that, they attached all those spike proteins to HIV-pseudoviruses and monitored how they would infect different cells. It is important to note, a pangolin spike protein was used as a control group because it contained no S1 & S2 segment and therefore didn’t need to be cleaved, or cut by TMPRSS2 like the original, Alpha, or Delta variants did.
Infecting other sets of cells with lab made HIV-psuedovirus having Alpha, Delta, Omicron, and pangolin like spike proteins on them(📸 FIG. 7)
Below, are other cells that were engineered to become infected by the previously mentioned HIV- pseudoviruses with specific spike proteins. From left to right below. The first cell, a CALU-3 CELL that has many TMPRSS2 and not many ACE2 receptors. The second cell, an A549 CELL that has both many TMPRSS2, and ACE2 receptors. The third cell, an HEK cell that has many ACE2 receptors, but minimal TMPRSS2. If you remember, TMPRSS2 enables Delta, original, and Alpha variants to infect a cell. However, Omicron skips this step and enters as an endocytotic package. That said here is the takeaway (from left to right):
HIV- pseudovirus with Delta spike protein attached easily and entered the CALU-3 CELL because there was an abundance of TMPRSS2 on the membrane. Conversely, HIV- pseudovirus with Omicron spike protein entered the cell much less because it had many less ACE2 receptors on the membrane.
Both HIV- pseudovirus with Delta spike proteins, and Omicron spike proteins entered the A549 cell equally because there was an abundance of both TMPRSS2 that Delta needs to enter the cell, and ACE2 receptors that Omicron needs to enter cell.
HIV- psuedovirus with Delta spike protein could not enter because there was minimal TMPRSS2. On the other hand, HIV- pseudovirus with Omicron spike protein entered easily because there was an abundance of ACE2 on the surface.
The takeaway 🔔: Since Omicron-like psuedoviruses easily entered cells with an abundance of ACE2 receptors, but did not enter as easily cells without, it could be said that Omicron enters then infects cells through endocytosis and therefore doesn’t use the same mechanism that Delta does. That just means Omicron is more infectious. Also, the HIV- pseudoviruses carrying anteater spike proteins with no S1 & S2 segment couldn’t infect any cells. That was used as a control to show that separation of S1 & S2 is essential for original, Alpha, and Delta variants to enter/infect cells.
Finally, just to confirm, scientists inhibited ALL PROTEASES on the surfaces of all 3 cells using a drug named CAMOSTAT (that includes TMPRSS2). Turns out, Omicron still infected the cells where proteases were inhibited because Omicron didn’t need TMPRSS2 protease, or any protease to enter like Delta needed. Furthermore, scientists then added E64D which inhibited cathepsin. The result, cathepsin couldn’t be liberated in Omicron so it couldn’t further infect the cells.
What does all of this mean?
Simply put, Omicron is more transmissible as it infects cells differently than did original, Alpha, and Delta variants. The nearly 50 mutations in Omicron allow it to enter and infect cells differently. The result, increased transmissibility but decreased severity of disease.
LETS CONNECT: